Why Understanding Defective Medical Devices Matters Now
Defective medical device claims affect thousands of Americans annually, causing serious injuries and even death. While medical devices are designed to save lives and improve health, their failure can be devastating.
Quick Answer: What You Need to Know
A medical device is considered defective if it causes harm due to design flaws, manufacturing errors, or inadequate warnings about its risks. If you’ve been harmed, you may be entitled to compensation for medical bills, lost wages, and pain and suffering.
The numbers are sobering. From 2008 to 2018, defective medical devices led to roughly 1.7 million injuries and 83,000 deaths in the U.S. With nearly 1 in 10 Americans having an implanted medical device, the risk is widespread.
These statistics represent real people who trusted a product to help them but instead faced chronic pain, additional surgeries, and life-altering complications. If you’re dealing with unexpected medical problems and mounting bills from a faulty device, know that you are not alone and you have options.
This guide explains how to identify a defective device, determine who is responsible, and take the necessary steps to protect your rights and pursue the compensation you may be owed.

What Is a Defective Medical Device?
A defective medical device is any medical instrument, implant, or piece of equipment that causes harm rather than providing the intended healing or support.
The U.S. Food and Drug Administration (FDA) regulates medical devices, but dangerous products still reach the market. A primary reason is the FDA’s 510(k) clearance process, which allows manufacturers to fast-track approval for a new device by claiming it is “substantially equivalent” to an existing one, often bypassing rigorous clinical trials.
Once a device is in use, the FDA tracks issues through its Medical Device Reporting (MDR) system, which receives hundreds of thousands of reports associated with medical devices annually. When a device is deemed dangerous, the FDA can issue a recall. The most serious are Class I recalls, which indicate a reasonable probability of serious injury or death. In 2023, there were 102 Class I recalls, showing that defective and recalled products remain a persistent threat.
The Three Types of Defects
When a defective medical device causes harm, the issue typically falls into one of three categories:
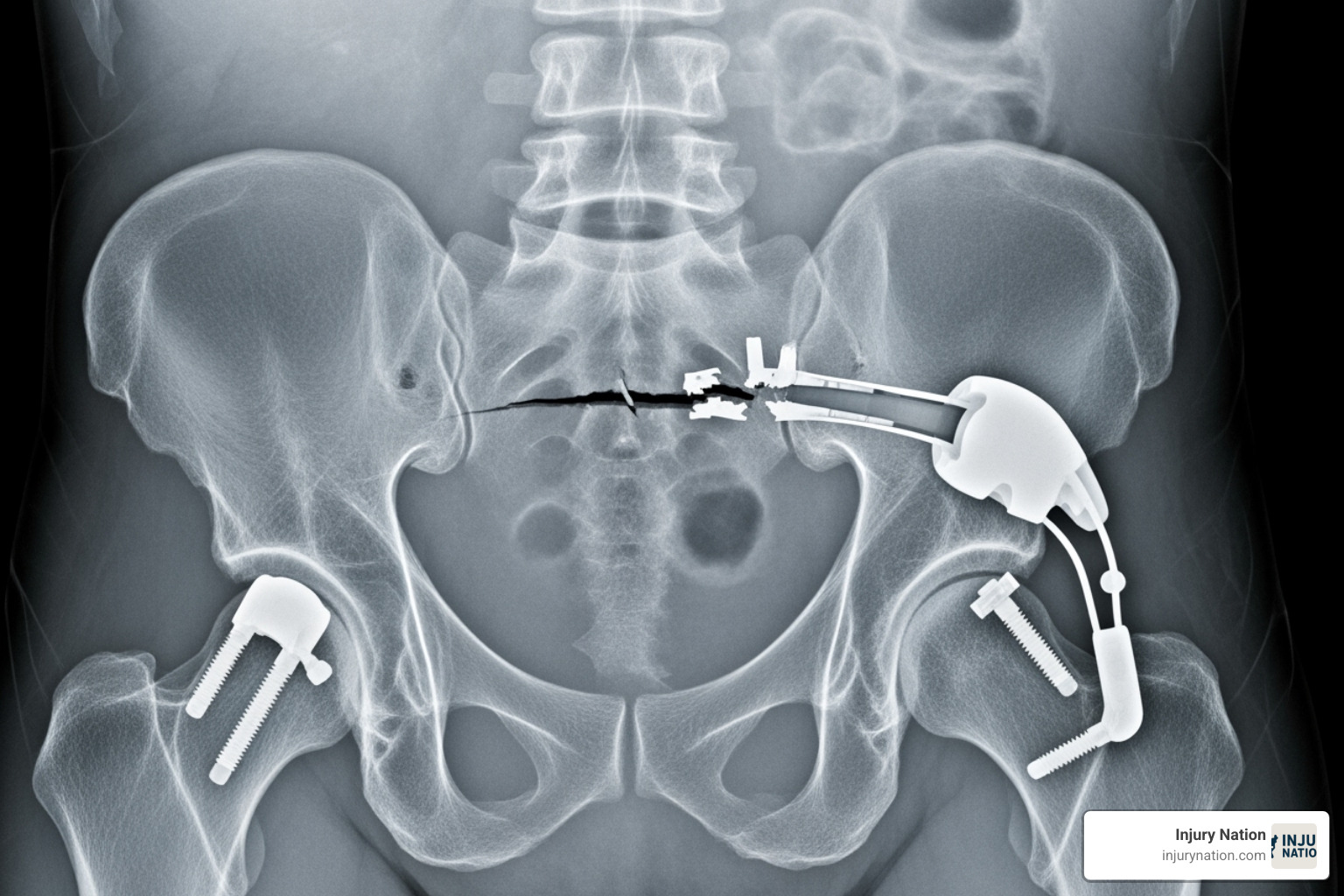
Design defects mean the device is inherently dangerous due to its fundamental design. Even with perfect manufacturing, the product is unsafe for its intended use. An example is a hip implant made from metals that corrode and release toxins.
Manufacturing defects occur when a sound design is compromised during production. This could be due to contamination, incorrect assembly, or other errors on the production line, affecting a single device or an entire batch.
Failure to warn (or marketing defects) happens when the manufacturer fails to provide adequate instructions or warn doctors and patients about known risks. Promoting a device for unapproved “off-label” uses also falls into this category.
Common Examples of a Defective Medical Device
Defective devices have appeared in nearly every field of medicine. Some common examples include:
- Hip and knee replacements (DePuy, Exactech) that fail prematurely, loosen, or cause metallosis (metal poisoning).
- Pacemakers and defibrillators that malfunction, failing to regulate the heartbeat correctly.
- Surgical mesh (Ethicon, C.R. Bard) used for hernia repair, which can erode, migrate, and cause chronic pain and organ damage.
- IVC filters designed to catch blood clots that fracture and perforate blood vessels.
- Insulin pumps that deliver incorrect doses of insulin.
- Contraceptive devices like Essure and certain IUDs that have caused organ perforation and chronic pain.
- Surgical tools, including surgical robots and staples, that malfunction and cause internal injuries.
While most medical devices are safe, these examples show the devastating consequences when they are not.
Identifying Harm and Establishing Liability
When a defective medical device causes injury, the consequences can be overwhelming, ranging from chronic pain to life-threatening complications.

Common health consequences include chronic pain, persistent infections, and the need for painful revision surgeries to remove or replace a failed device. In some cases, devices can break apart or corrode, causing organ damage. The most tragic outcome is wrongful death. High-profile examples include metal-on-metal hip implants causing metallosis and pelvic mesh devices eroding through tissue, causing debilitating pain.
It’s important to distinguish a defective medical device claim from medical malpractice. A defective device claim focuses on the product itself, while malpractice involves a healthcare provider’s negligence. However, sometimes both issues are present in a single case.
Who Can Be Held Liable?
Multiple parties in the device’s chain of distribution can be held responsible for injuries, including:
- Manufacturers who designed, produced, or tested the device.
- Designers if they created an inherently unsafe product.
- Testing labs that failed to conduct adequate safety evaluations.
- Distributors and sales representatives who promoted the device for unapproved uses or failed to warn of known defects.
- Hospitals and healthcare providers, typically under a theory of negligence if they implanted a device incorrectly or failed to monitor a patient properly.
The Legal Basis for a Defective Medical Device Claim
Most lawsuits are based on product liability law, often using a legal principle called strict liability. Under this standard, you don’t have to prove the manufacturer was negligent. Instead, you must show that the device was defective, the defect existed when it left the manufacturer, and the defect directly caused your injuries. This is what lawyers call proving a prima facie case.
Causation is often the most challenging element to prove, requiring a clear link between the defect and your specific harm. This is established with extensive medical records and testimony from expert witnesses, such as medical and engineering specialists.
Be aware of the statute of limitations, a strict deadline for filing a lawsuit that varies by state. If you miss it, you lose your right to seek compensation.
When a device harms many people, lawsuits may be consolidated. Class actions group many victims into a single lawsuit. Multidistrict litigation (MDL) consolidates similar cases before one judge for pretrial proceedings, which helps streamline the process, though each case remains individual.
Your Step-by-Step Guide to Filing a Defective Medical Device Claim
Finding out that a medical device meant to improve your health has instead caused harm is devastating. You’re probably feeling overwhelmed, frustrated, and uncertain about what to do next. But here’s what you need to know: you don’t have to steer this alone, and taking the right steps now can make all the difference in protecting your rights.
Time is critical when it comes to defective medical device claims. Every state has a statute of limitations—a legal deadline for filing your lawsuit. If you miss this window, you could lose your right to seek compensation entirely. That’s why acting promptly matters so much.

Step 1: Seek Medical Attention and Document Everything
Your health is the top priority. See a doctor immediately to diagnose the problem. If the device is removed, ask your medical team to preserve it, as it is critical evidence.
Start a journal to document your symptoms, pain levels, and how the injury affects your daily life. Gather all related paperwork, including medical records, test results, bills, and any communication from the manufacturer or your doctors about the device.
Step 2: Report the Issue
Reporting the problem helps your case and protects others. You can file a voluntary report with the FDA through its MedWatch Safety Information and Adverse Event Reporting Program. You can also search the FDA’s Manufacturer and User Facility Device Experience (MAUDE) database to see if others have reported similar issues with the same device.
Step 3: Consult with a Product Liability Attorney
Defective medical device cases are incredibly complex, and you will be facing large corporations with powerful legal teams. An experienced product liability attorney is essential.
Your attorney will investigate your claim, hire experts to prove what went wrong, identify all liable parties, and handle all negotiations. They understand the tactics used by defense lawyers and insurance companies to deny or reduce claims and can effectively counter them. Most work on a contingency fee basis, meaning you pay nothing unless they win your case.
Step 4: Understand Potential Compensation
If your claim is successful, you may be entitled to several types of compensation.
- Economic damages cover financial losses like past and future medical bills, lost wages, and diminished earning capacity.
- Non-economic damages compensate for non-financial losses, such as pain and suffering, emotional distress, and loss of enjoyment of life.
- Punitive damages may be awarded in rare cases to punish a manufacturer for particularly reckless or malicious behavior, such as knowingly hiding a device’s dangers.
In the tragic event of a death, surviving family members may file a wrongful death claim to cover funeral expenses and loss of financial and emotional support.
Frequently Asked Questions about Defective Medical Device Claims
If you’re dealing with a potentially defective medical device, you’re probably feeling overwhelmed and uncertain about what comes next. We’ve heard these concerns countless times, and we want to address the questions that matter most to you right now.
How much does it cost to hire a lawyer for a defective medical device case?
Most product liability attorneys work on a contingency fee basis. This means you pay no upfront fees, and the lawyer only gets paid a percentage of the compensation they recover for you. If you don’t win your case, you owe no attorney’s fees. Most firms also offer a free initial consultation to discuss your case without any obligation. This system allows anyone who has been harmed to seek justice, regardless of their financial situation.
How long does a defective medical device lawsuit take?
The timeline for a defective medical device lawsuit varies greatly. Straightforward cases may resolve in a year or two, while more complex litigation can take several years. Factors influencing the timeline include the complexity of the medical evidence, whether the case is individual or part of a larger multidistrict litigation (MDL), and whether the case settles or goes to trial. Many cases are resolved through settlement negotiations, which can be faster than a full trial.
Can I pursue a claim if my device was recalled but I haven’t been injured yet?
Generally, you must have suffered an actual injury to file a personal injury claim for damages. If a device was recalled but you have no symptoms, you typically cannot sue for compensation for an injury that hasn’t happened.
However, some class action lawsuits may establish medical monitoring programs, which cover the cost of future health screenings to detect problems early. If you do develop injuries later, you would then have grounds for a claim. It is wise to consult an attorney even if you are not yet injured to understand your rights and the applicable statute of limitations.
Take the Next Step: Find Legal Support for Your Claim

You trusted a medical device to improve your health, but instead, it caused harm. This is a betrayal of trust, and you have the right to seek justice. Holding manufacturers accountable not only helps you but also pushes the industry to prioritize patient safety.
These cases are too complex to handle alone. Medical device companies have vast resources to defend themselves. You need an expert on your side. An attorney specializing in defective medical device cases knows how to build a strong claim, steer the legal system, and fight for the compensation you deserve.
We created Injury Nation specifically to help people like you connect with these specialized attorneys. We’ve built a network of top-rated Personal Injury Lawyers across America who have proven track records in handling complex medical device cases. Our goal is simple: to make it easy for you to find an attorney who understands your situation and can fight effectively for the compensation you deserve.
The path forward starts with a conversation. Most attorneys in our network offer free consultations, giving you the opportunity to discuss your case without any financial commitment. They’ll review your situation, explain your legal options, and help you understand what to expect. There’s no pressure, no obligation—just honest guidance from someone who knows this area of law inside and out.
Time is critical here. Remember those statutes of limitations we discussed? The clock is ticking on your right to file a claim. The sooner you connect with an attorney, the sooner they can begin preserving evidence, documenting your injuries, and building your case.
Don’t steer this complex process alone. You’re already dealing with enough—the physical pain, the emotional stress, the financial burden. Let an experienced attorney handle the legal battle while you focus on your recovery and your family.
Visit Injury Nation today to connect with a qualified attorney in your area who can evaluate your case and fight for the justice and compensation you deserve. Your journey toward recovery and accountability starts with that first step.



